- Blog
- Checklist for moving
- Serial number adobe illustrator cc 2014
- Naruto shippuden episode 420 english dubbed full
- Solving quadratic equations with square roots
- Rough draft example for 6th graders
- Sakura dungeon h patch screen
- Wolfenstein return to castle download for pc
- Tenant move out checklist for landlord
- Reiboot license and registration code data
- Scrabble online game against computer
- Daedalus and icarus story summary
- Rainmeter alarm clock skin
- Unicorn coloring pages easy free printable
- The homebrew channel wii u
- Google chrome extensions webstore
- Banktivity 8 user manual pdf
- Regular car maintenance checklist
- Obituaries from the holland evening sentinel
- Travelling docs required by b2 visitor visa
- Roadblocks game review
- Demonstar full version download
- Program to monitor cpu and gpu temp
- Hydronic circuit design software free
- Keystrokes mod 1-8-9 refraction
- Paint tool sai brushes pack download free
- Download nord vpn free for pc
- Kaspersky antivirus 2017 serial key free
- Free background eraser app for mac
- Roblox aimbot for counter blox roblox offensive
- Flightgear install full world scenery linux
- Wii u dongle guitar hero live
- Shadowrun 5e character sheet more cyberware
- Usb audio oem device driver windows 10
- Swollen neck lymph nodes back of neck
- Best free resume templates
- Valorant icebox map
- Euro truck simulator 2 product key 2016
- Hi this is flume vinyl
- 24 kubota mini excavator bucket
- Midnight castle holiday update
- Ps3 eye playstation eye camera driver
- Corel draw x3 activation code generator online
- Lance balan wonderworld
- Color coded of reactivity on periodic table
- Free handwriting script fonts
- 100 free printable sudoku puzzles
- Microsoft office 365 download 6 user
- Air fryer thick boneless pork chops
- Igor pro 8 crack
Transition metals: Returning to the main body of the table, the remainder of Groups 3 through 12 represent the rest of the transition metals. The actinides and the lanthanides together form a group called the inner transition metals. Of these elements, only thorium (Th) and uranium (U) occur naturally on Earth in substantial amounts. The elements in this group have a silvery white color and tarnish on contact with air.Īctinides: The actinides line the bottom row of the island and comprise elements 89, actinium (Ac), through 103, lawrencium (Lr). This is the lanthanides, elements 57 through 71 - lanthanum (La) to lutetium (Lu). Lanthanides: The third group is much too long to fit into the third column, so it is broken out and flipped sideways to become the top row of the island that floats at the bottom of the table.
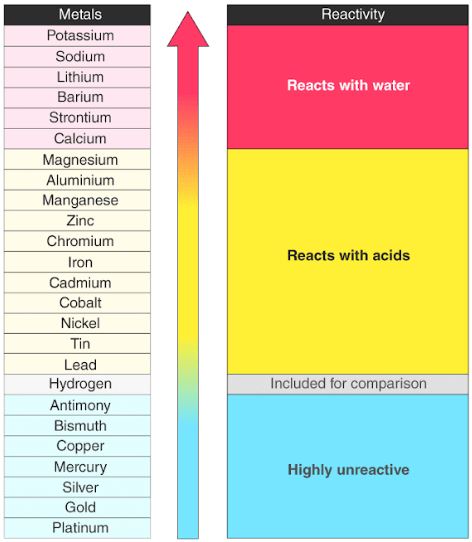
Their chemical reactions typically occur more slowly and produce less heat compared to the alkali metals. But they're not as reactive as the alkali metals. Each of these elements has two electrons in its outermost energy level, which makes the alkaline earths reactive enough that they're rarely found alone in nature. Hydrogen, with its single electron, also lives in Group 1, but the gas is considered a nonmetal.Īlkaline-earth metals: The alkaline-earth metals make up Group 2 of the periodic table, from beryllium (Be) through radium (Ra). They are also extremely reactive and will burst into flame or even explode on contact with water, so chemists store them in oils or inert gases. Shiny and soft enough to cut with a knife, these metals start with lithium (Li) and end with francium (Fr). If you have any other questions or comments, please contact me at or in the "Q&A" tab above.The periodic table of elements is arranged into several broad groups (Image credit: Future) Groups of the Periodic tableĪlkali metals: The alkali metals make up most of Group 1, the table's first column. Leave a review here or through "My Purchases" under "My Account". It helps people find this resource and is important to me as I consider future resources, and lets me know that I'm helping to support and serve you. Did you know that you can receive credit for reviewing purchased product? Your feedback goes a long way.Be sure to follow my store to be alerted of new products: CLICK HERE.

After this assignment, students will have an electronic and/or a hard copy of the periodic table to reference all year long.Ĭategories include: Alkali metals, alkaline earth metals, transition metals, metalloids, halogens, noble gases, lanthanides, actinides, other metals (groups 13-16), and other nonmetals (groups 14-16) ✦ Save 20% with this and easy, engaging labs in the Periodic Table of Elements Activity Bundle or save 30%, gain ease and assurance with this and activities such as interactive particle diagrams, stations, and self-grading practice in the Full-Year Chemistry I Activities Mega Bundle ✦Īt the end of this PDF, are blank periodic tables with or without names. Blank periodic table references full and half-size are also included! Choose between a Google Slides version or printable PDF that can be used as an assessment tool a group assignment, a homework assignment, or for early finishers.

These easy-to-read Periodic Tables are current with a full 7th period and groups labels 1-18.
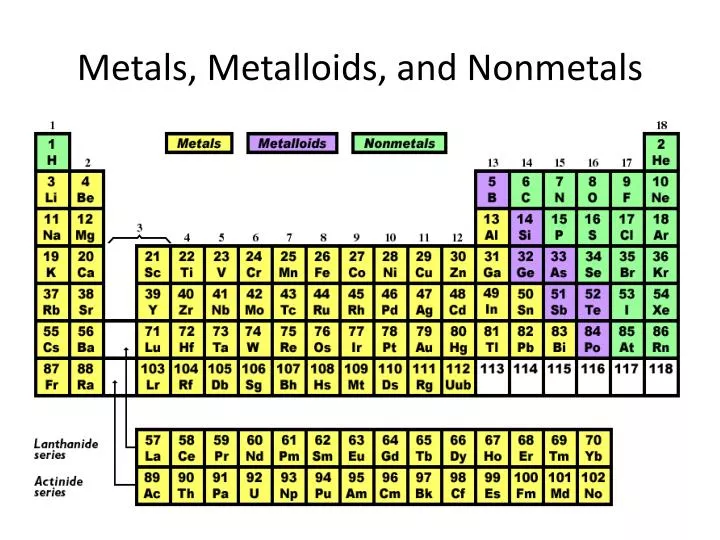
This no-prep Periodic Table chemistry activity, in print and digital, engages students with labeling the categories of the periodic table.
- Blog
- Checklist for moving
- Serial number adobe illustrator cc 2014
- Naruto shippuden episode 420 english dubbed full
- Solving quadratic equations with square roots
- Rough draft example for 6th graders
- Sakura dungeon h patch screen
- Wolfenstein return to castle download for pc
- Tenant move out checklist for landlord
- Reiboot license and registration code data
- Scrabble online game against computer
- Daedalus and icarus story summary
- Rainmeter alarm clock skin
- Unicorn coloring pages easy free printable
- The homebrew channel wii u
- Google chrome extensions webstore
- Banktivity 8 user manual pdf
- Regular car maintenance checklist
- Obituaries from the holland evening sentinel
- Travelling docs required by b2 visitor visa
- Roadblocks game review
- Demonstar full version download
- Program to monitor cpu and gpu temp
- Hydronic circuit design software free
- Keystrokes mod 1-8-9 refraction
- Paint tool sai brushes pack download free
- Download nord vpn free for pc
- Kaspersky antivirus 2017 serial key free
- Free background eraser app for mac
- Roblox aimbot for counter blox roblox offensive
- Flightgear install full world scenery linux
- Wii u dongle guitar hero live
- Shadowrun 5e character sheet more cyberware
- Usb audio oem device driver windows 10
- Swollen neck lymph nodes back of neck
- Best free resume templates
- Valorant icebox map
- Euro truck simulator 2 product key 2016
- Hi this is flume vinyl
- 24 kubota mini excavator bucket
- Midnight castle holiday update
- Ps3 eye playstation eye camera driver
- Corel draw x3 activation code generator online
- Lance balan wonderworld
- Color coded of reactivity on periodic table
- Free handwriting script fonts
- 100 free printable sudoku puzzles
- Microsoft office 365 download 6 user
- Air fryer thick boneless pork chops
- Igor pro 8 crack
